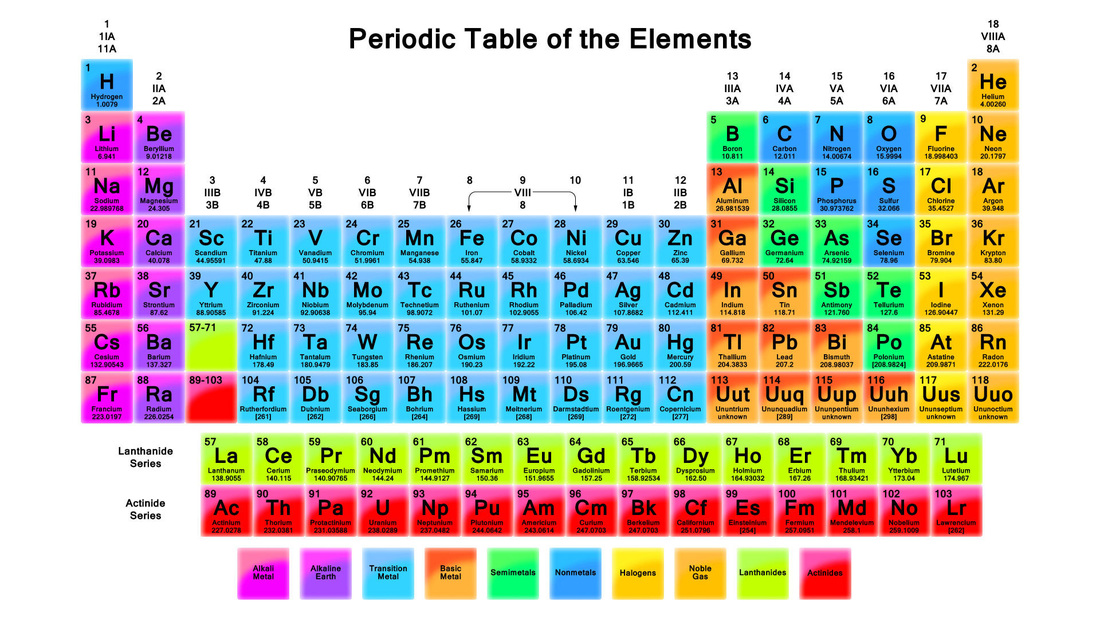
The Russian chemist Dmitri Mendeleev was the first scientist to make a periodic table similar to the one used today. His periodic table was published in 1896, and was widely used. Mendeleev arranged the elements by atomic mass, corresponding to relative molar mass (the mass of a given substance divided by its amount of substance). The arrangement of the elements, or of groups of elements in the order of their atomic masses, corresponds to their valencies, as well as to their distinctive chemical properties. The only two problems with Mendeleev's periodic table is that it didn't include any of the noble gases, and that it didn't have a set position for hydrogen.
Later on, a scientist by the name of Glenn T. Seaborg made some modifications to Mendeleev's table: he added the Actinide series. He had experienced unexpected difficulties in isolating the elements americium and curium, so he wondered if these elements belonged to a different series. This is what influenced his decision to add the Actinide series to the table.
Later on, a scientist by the name of Glenn T. Seaborg made some modifications to Mendeleev's table: he added the Actinide series. He had experienced unexpected difficulties in isolating the elements americium and curium, so he wondered if these elements belonged to a different series. This is what influenced his decision to add the Actinide series to the table.